Surgical forceps are among the most essential tools in modern healthcare, playing a critical role in everything from routine wound dressing to complex surgical procedures. For clinicians, having access to forceps that combine precision, durability, and compliance with global standards is non-negotiable—these tools directly impact patient outcomes and the efficiency of healthcare delivery. In this article, we explore the key features of a leading line of surgical forceps, their advantages over competitors, the advanced manufacturing processes that set them apart, and how they address the evolving needs of healthcare providers worldwide.
What Are Surgical Forceps? A Comprehensive Overview
Surgical forceps are handheld instruments designed to grasp, hold, or manipulate tissues, dressings, or other objects during medical procedures. They are categorized based on their intended use, with each type featuring unique jaw designs, sizes, and functionalities. The line of forceps discussed in this article includes four key models, each tailored to specific clinical needs:
- Dressing Forceps (SMD-131801): Used for applying, removing, or adjusting wound dressings. Their smooth jaws prevent tissue damage while providing a secure grip on gauze, bandages, or other dressing materials.
- Tissue Forceps (SMD-131802): Designed for grasping and holding biological tissues during surgical procedures. These forceps often have serrated or textured jaws to ensure a firm hold without slipping, making them ideal for suturing or tissue manipulation.
- Anatomise Forceps (SMD-131803): A specialized tool used for precise dissection and separation of tissues. Their narrow, pointed jaws allow clinicians to work in tight spaces, making them essential for delicate anatomical procedures.
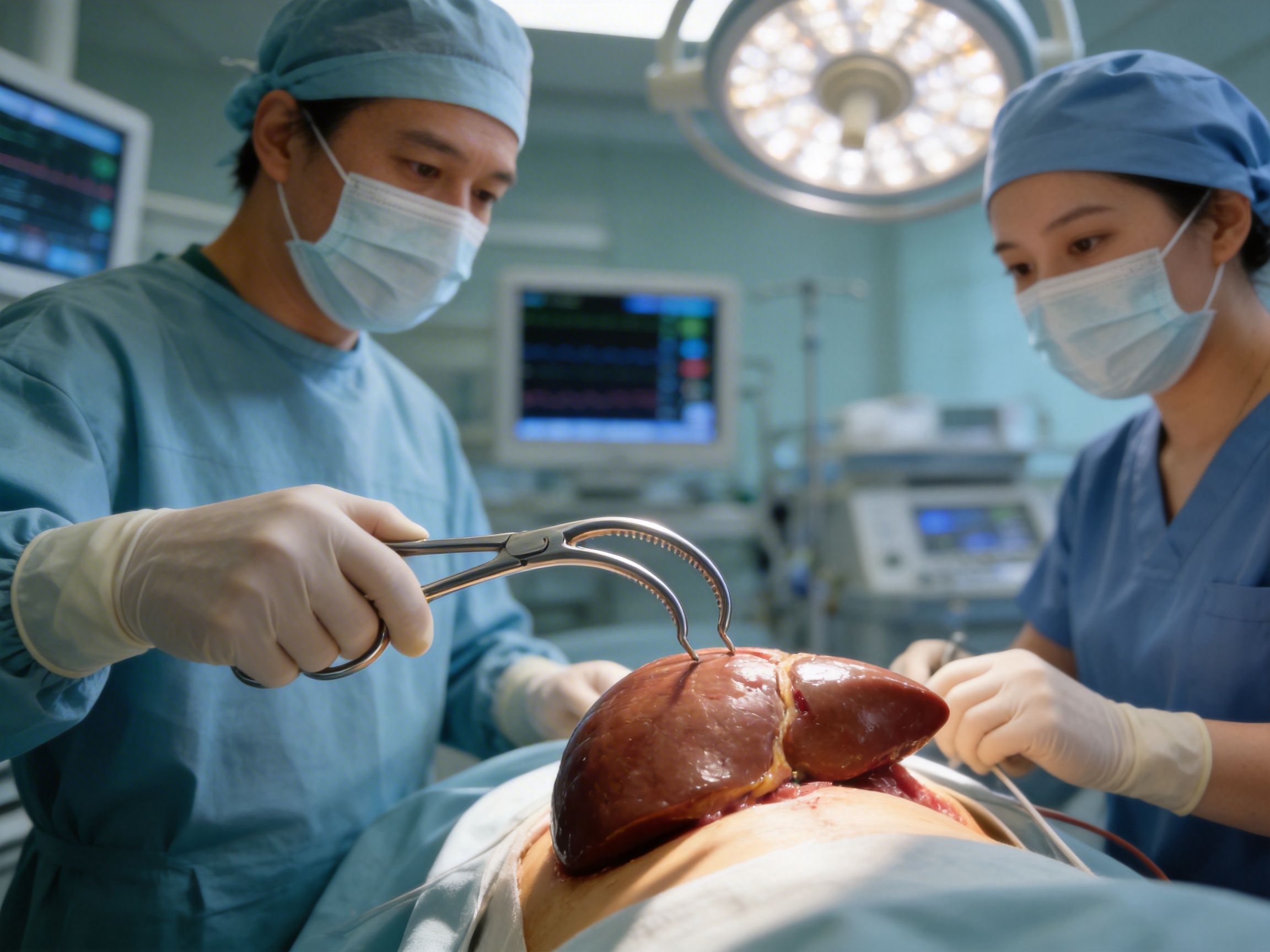
- Surgical Hepatic Forceps (SMD-131804): Tailored for use in hepatic (liver) surgery. These forceps feature curved jaws and a design that minimizes trauma to liver tissue, enabling surgeons to handle delicate hepatic structures with precision.
Each of these forceps models is available in multiple size options, ensuring that clinicians can select the exact tool needed for each procedure—whether it’s a small 12.5cm forceps for a minor wound or a 25cm model for deep-tissue access.
Key Advantages of the Featured Surgical Forceps
The line of surgical forceps discussed here stands out in the medical device market due to a combination of design, material quality, and compliance. Below are the core advantages that make these forceps a preferred choice for healthcare providers:
1. Precision Engineering for Clinical Excellence
Precision is the cornerstone of any high-quality surgical instrument, and these forceps are no exception. Each model is manufactured with micrometer-level accuracy, ensuring consistent jaw alignment, grip strength, and overall performance. For example, the tissue forceps feature serrated jaws that are precisely cut to provide a secure hold without tearing delicate tissues—a critical feature for surgeons performing suturing or tissue resection. The dressing forceps, on the other hand, have smooth, rounded jaws that prevent accidental tissue damage while still holding dressings firmly in place. This level of precision reduces the risk of procedural errors and improves patient safety.
2. Unmatched Versatility Across Clinical Settings
One of the most notable advantages of these forceps is their versatility. With multiple size options for each model, clinicians can adapt the tool to the specific needs of their patients and procedures. For instance, a 12.5cm anatomise forceps is ideal for ophthalmic or ENT procedures where space is limited, while a 25cm dressing forceps can be used for wound care on larger areas of the body (e.g., post-operative incisions on the abdomen). This versatility eliminates the need for clinicians to use multiple tools for similar tasks, streamlining workflow and reducing the risk of tool mix-ups.
3. Compliance with Global Regulatory Standards
Healthcare providers around the world rely on regulatory certifications to ensure that medical devices meet safety and performance requirements. These forceps are certified to meet three key global standards:
- ISO 13485:2016: This international standard specifies requirements for a quality management system (QMS) for medical devices. Compliance ensures that the manufacturing process is consistent, traceable, and focused on patient safety.
- CE Marking: Required for medical devices sold in the European Union, this certification confirms that the forceps meet the safety, health, and environmental protection requirements of the EU Medical Device Regulation (MDR 2017/745).
- FDA Registration: The U.S. Food and Drug Administration (FDA) has registered these forceps, meaning they comply with the FDA’s Quality System Regulation (21 CFR Part 820) and are safe for use in the U.S. healthcare market.
These certifications mean that the forceps can be used in over 60 countries worldwide, including key markets in Europe, Southeast Asia, Africa, and Oceania—without the need for additional regulatory approvals in most cases.
4. Cost-Effectiveness Without Compromising Quality
Healthcare facilities face constant pressure to manage costs while maintaining high standards of care. These forceps strike a balance between quality and affordability, making them an ideal choice for hospitals, clinics, and first aid suppliers. Unlike some premium-priced competitors that offer similar features, these forceps are priced competitively due to the manufacturer’s efficient production processes and 30 years of industry experience. This cost-effectiveness does not come at the expense of quality—each forceps undergoes rigorous testing to ensure it meets or exceeds global standards.
Advanced Manufacturing Processes Behind the Forceps
The exceptional quality of these surgical forceps is a direct result of the manufacturer’s advanced manufacturing processes and commitment to continuous improvement. Below is a detailed look at the key stages of production:
1. Material Sourcing and Validation
The manufacturing process begins with the sourcing of high-grade 304 stainless steel— a material chosen for its unique combination of strength, corrosion resistance, and biocompatibility. 304 stainless steel is widely used in medical instruments because it can withstand repeated sterilization cycles (autoclaving) without rusting or degrading, and it does not react with human tissue. The manufacturer sources raw steel from certified suppliers and conducts rigorous validation tests to ensure it meets the required specifications (e.g., chemical composition, tensile strength).
Each batch of raw material is assigned a unique traceability code, which is tracked throughout the entire production process. This traceability ensures that if any issue is identified in a finished product, the manufacturer can quickly identify the source of the problem and take corrective action.
2. Precision Machining and Finishing
Once the raw material is validated, it is sent to the machining department, where computer numerical control (CNC) machines are used to shape the forceps. CNC machining ensures that each forceps is manufactured to exact specifications, with micrometer-level accuracy. For example, the jaws of the tissue forceps are machined to have precise serrations that are uniform across all units—this consistency is critical for ensuring that each forceps performs the same way, regardless of the batch.
After machining, the forceps undergo a series of finishing processes:
- Polishing: The forceps are polished to remove any burrs or rough edges, which could cause tissue damage or interfere with the tool’s functionality.
- Passivation: A chemical process that creates a protective oxide layer on the surface of the stainless steel, enhancing its corrosion resistance.
- Sterilization Testing: The forceps are tested for compatibility with common sterilization methods to ensure they can withstand repeated cycles without degradation.
3. Rigorous Quality Control Protocols
Quality control is integrated into every stage of the manufacturing process, with hundreds of dedicated QC personnel overseeing inspections. Key quality control steps include:
- In-Process Inspections: At each stage of production (machining, finishing, assembly), inspectors check the forceps for dimensional accuracy, surface finish, and functionality.
- Functional Testing: Each forceps is tested to ensure it opens and closes smoothly, the jaws align perfectly, and the grip strength meets the required specifications. For example, tissue forceps are tested to ensure they can hold a 500g weight without slipping.
- Final Inspection: Before shipping, each batch of forceps undergoes a final inspection to confirm that it meets all regulatory standards and customer requirements. A certificate of compliance is issued for each batch.
The manufacturer’s commitment to quality is further demonstrated by its adherence to ISO 13485:2016, which requires regular internal and external audits to ensure the QMS is effective.
4. Customization Capabilities
In addition to standard models, the manufacturer offers OEM (Original Equipment Manufacturer) services that allow clients to customize forceps to their specific needs. Customization options include:
- Custom Sizes: Clients can request size variations beyond the standard options listed (e.g., a 10cm anatomise forceps for pediatric procedures).
- Branding: Clients can add their logo, facility name, or inventory code to the forceps for easy identification and traceability.
- Specialized Features: For clients with unique clinical needs, the manufacturer can design and manufacture forceps with specialized features (e.g., curved jaws for hard-to-reach areas).
These customization options make the forceps suitable for a wide range of healthcare settings, from large hospitals to specialized clinics.
How These Forceps Outperform Competitors
In a crowded medical device market, these forceps stand out due to their combination of features, quality, and value. Below are the key ways they outperform their competitors:
1. Greater Size Variety for Clinical Flexibility
One of the most significant advantages of these forceps is their extensive size range. For example, the dressing forceps are available in six sizes (12.5cm to 25cm), while many competitors only offer three or four sizes. This greater variety means that clinicians can select the exact size needed for each procedure—whether it’s a small forceps for a minor wound or a large one for deep-tissue access. Competitors often force clinicians to compromise on size, which can lead to reduced efficiency or increased risk of tissue damage.
2. Consistent Quality Across Batches
Consistency is a critical factor in medical instrument manufacturing, as clinicians rely on tools that perform the same way every time. The manufacturer’s 30 years of industry experience and rigorous quality control processes ensure that each batch of forceps is identical in terms of quality and performance. In contrast, many competitors (especially smaller manufacturers) struggle with batch-to-batch consistency due to outdated manufacturing processes or insufficient QC resources. This inconsistency can lead to unexpected tool failures during procedures, which pose a risk to patient safety.
3. Comprehensive Global Regulatory Compliance
While many competitors offer forceps that meet one or two regulatory standards, these forceps comply with three key global standards (ISO 13485, CE, FDA). This comprehensive compliance means that the forceps can be used in almost any country without additional approvals—an important advantage for healthcare providers that operate in multiple markets or export their services. Competitors that lack FDA registration, for example, cannot sell their forceps in the U.S. market, limiting their reach.
4. End-to-End Support from Experts
The manufacturer’s 30 years of experience in the medical device industry means that it can provide end-to-end support to clients—from helping them select the right forceps for their needs to offering after-sales service. For example, the manufacturer’s team of experts can advise clients on the best sterilization methods for the forceps, or help them design custom forceps for specialized procedures. Many competitors lack this level of expertise, leaving clients to figure out these issues on their own.
Case Study: Real-World Application in a European Hospital
To illustrate the impact of these forceps in clinical settings, let’s look at a case study of a large hospital in Germany that has been using these forceps for the past two years.
Background
The hospital is a 500-bed facility that serves a diverse patient population, including emergency cases, surgical patients, and outpatients. Prior to switching to these forceps, the hospital used a mix of forceps from three different suppliers, which led to inconsistent quality, limited size options, and higher costs.
Implementation
The hospital’s procurement team selected these forceps based on their comprehensive regulatory compliance, extensive size range, and competitive pricing. The team worked with the manufacturer to customize some of the forceps (e.g., adding the hospital’s logo for inventory management) and received training on the best practices for using and sterilizing the tools.
Results
Since implementing these forceps, the hospital has seen several key improvements:
- Reduced Procedural Errors: The consistent quality and precision of the forceps have reduced the number of procedural errors related to tool functionality (e.g., slipping jaws, tissue damage). For example, the number of wound dressing errors decreased by 15% in the first year of use.
- Improved Efficiency: The extensive size range means that clinicians no longer have to search for the right tool—they can quickly select the size needed for each procedure. This has reduced the average time for wound dressing procedures by 10%.
- Lower Costs: The competitive pricing of the forceps, combined with their durability (they last 20% longer than the previous supplier’s tools), has reduced the hospital’s annual spending on surgical forceps by 22%.
- Positive Staff Feedback: Clinicians have praised the forceps for their comfort, precision, and reliability. A survey of 50 nurses and surgeons found that 92% preferred these forceps over their previous tools.
Quotes from hospital staff highlight the impact of the forceps:
- “The dressing forceps we use now have every size we need—no more running to the storage room to find the right tool. This saves us time and reduces the risk of delays in patient care.” — Nurse Anna, Emergency Department
- “The tissue forceps’ precision is unmatched. I can suture delicate tissues with confidence knowing I won’t tear them. This has improved patient outcomes in our surgical unit.” — Dr. Markus, General Surgeon
- “The hepatic forceps are perfect for our liver surgery cases. The curved jaws and 22cm size allow us to access deep areas without causing unnecessary trauma. We’ve had no issues with these forceps since we started using them.” — Dr. Lena, Hepatic Surgeon
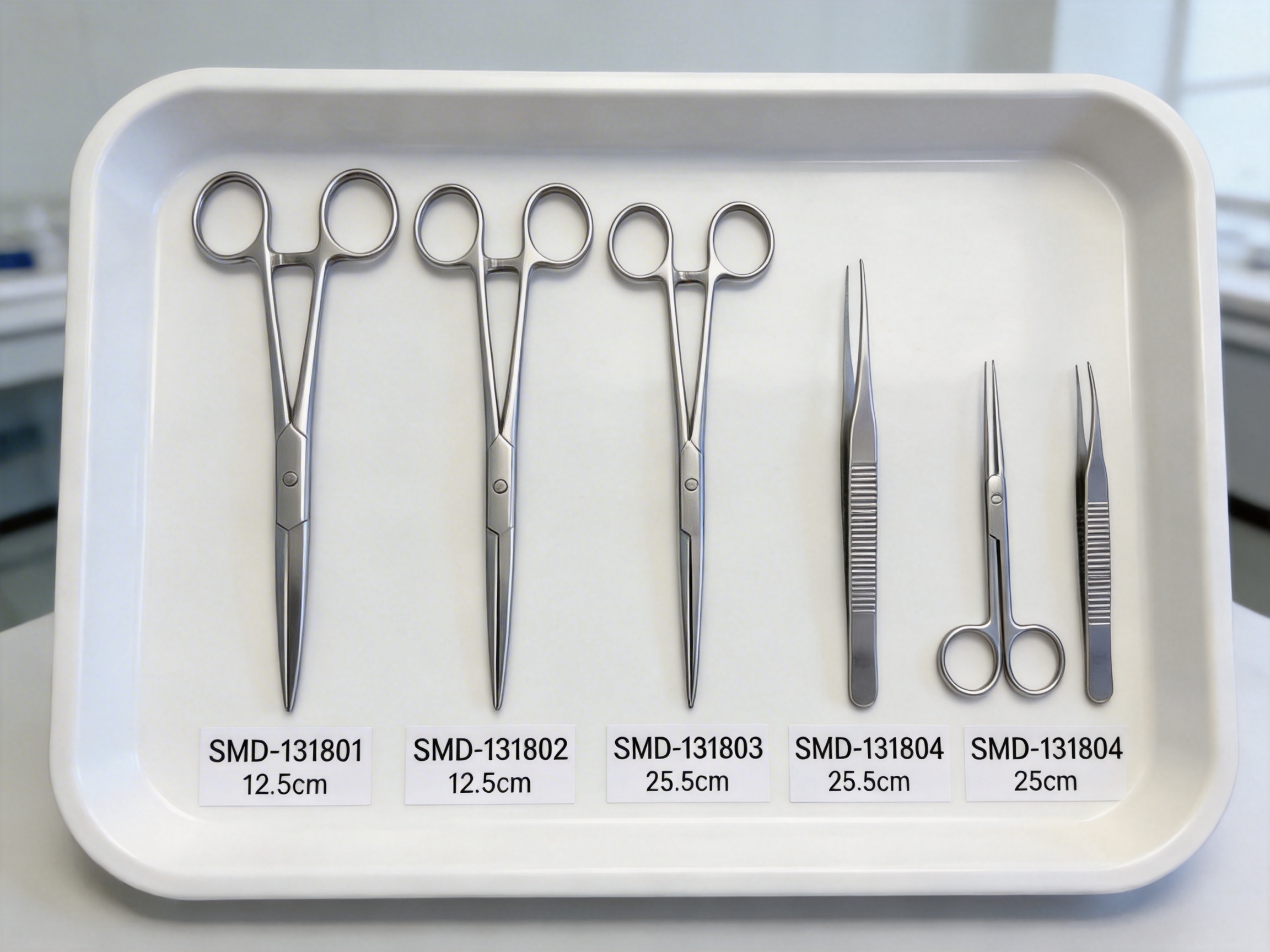
Table: Comparison of Featured Forceps vs. Competitor Models
| Forceps Type |
Our Model (Size Options) |
Competitor A (Size Options) |
Competitor B (Size Options) |
Certifications |
Primary Material |
| Dressing Forceps |
SMD-131801 (12.5cm,14cm,16cm,18cm,20cm,25cm) |
Model X (14cm,16cm,18cm) |
Model Y (12.5cm,16cm,20cm) |
ISO13485, CE, FDA |
High-grade Stainless Steel (304) |
| Tissue Forceps |
SMD-131802 (12.5cm,14cm,16cm,18cm,20cm,25cm) |
Model X (14cm,18cm,25cm) |
Model Y (16cm,20cm) |
ISO13485, CE, FDA |
High-grade Stainless Steel (304) |
| Anatomise Forceps |
SMD-131803 (12.5cm) |
Model X (12.5cm only) |
Model Y (10cm,12.5cm) |
ISO13485, CE, FDA |
High-grade Stainless Steel (304) |
| Surgical Hepatic Forceps |
SMD-131804 (12.5cm,14cm,16cm,18cm,20cm,22cm) |
Model X (16cm,18cm,22cm) |
Model Y (14cm,20cm,22cm) |
ISO13485, CE, FDA |
High-grade Stainless Steel (304) |
This table clearly shows the advantages of the featured forceps over their competitors, including a wider size range, more comprehensive certifications, and the use of high-grade 304 stainless steel.
Q&A Section
To address common questions about these surgical forceps, we’ve compiled the following Q&A:
Q1: What materials are used in these surgical forceps?
A1: These forceps are manufactured from high-grade 304 stainless steel. This material is chosen for its exceptional corrosion resistance, strength, and biocompatibility—making it ideal for medical instruments that need to withstand repeated sterilization cycles and contact with human tissue.
Q2: Are these forceps compatible with standard sterilization methods?
A2: Yes. The 304 stainless steel construction is fully compatible with all common medical sterilization techniques, including autoclaving (steam sterilization), dry heat sterilization, and ethylene oxide (EtO) sterilization. This ensures that the forceps can be safely reused across multiple procedures while adhering to infection control guidelines.
Q3: Can I order custom sizes or branding for these forceps?
A3: Absolutely. The manufacturer offers OEM (Original Equipment Manufacturer) services that allow for custom size adjustments and branding. Clients can request specific size variations beyond the standard options, as well as add their logo or facility name to the forceps for easy identification and inventory management.
Q4: What global certifications do these forceps hold?
A4: These forceps comply with three key international regulatory standards: ISO 13485:2016 (Quality Management System for Medical Devices), CE marking (European Union Medical Device Regulation), and FDA registration (U.S. Food and Drug Administration). These certifications ensure the forceps meet the highest safety and performance requirements for use in global healthcare markets.
Q5: How long is the lead time for bulk orders?
A5: Lead times vary based on order volume and customization requests. For standard orders (non-customized, in-stock sizes), lead times typically range from 15 to 25 business days. For custom orders (OEM branding, custom sizes), lead times may extend to 30 to 45 business days to accommodate design, production, and quality control processes.
Q6: What is the warranty period for these forceps?
A6: The manufacturer offers a 12-month warranty for these forceps, covering defects in materials and workmanship. If a forceps fails to perform as expected within this period, the manufacturer will replace it free of charge.
References
1. International Organization for Standardization. ISO 13485:2016: Medical devices—Quality management systems—Requirements for regulatory purposes. Geneva, Switzerland: ISO; 2016.
2. U.S. Food and Drug Administration. 21 CFR Part 820: Quality system regulation. Washington, DC: FDA; 2023.
3. European Commission. Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices. Official Journal of the European Union; 2017.
4. World Health Organization. Guidelines for infection prevention and control in healthcare settings. Geneva, Switzerland: WHO; 2020.
5. Journal of Medical Devices. Precision machining in surgical instrument manufacturing: A review of current practices and future trends. Vol. 15, Issue 2; 2022: 123-145.
6. Medical Device & Diagnostic Industry. The role of 304 stainless steel in medical instrument design. Vol. 45, Issue 3; 2021: 67-72.

 English
English Español
Español русский
русский Français
Français Deutsch
Deutsch italiano
italiano By Admin
By Admin