Introduction
The global wound care landscape is evolving rapidly, driven by an aging population, rising prevalence of chronic wounds (such as diabetic foot ulcers and pressure injuries), and increasing demand for safe, effective medical consumables. According to a 2023 report by Grand View Research, the global wound care market was valued at USD 22.5 billion and is projected to grow at a compound annual growth rate (CAGR) of 5.3% through 2030. At the heart of this market are abdominal pads—versatile dressings that play a critical role in post-operative care, emergency trauma management, and chronic wound treatment. However, not all abdominal pads are created equal: many generic products lack the absorbency, skin safety, and usability needed to optimize patient outcomes. This article explores a next-generation abdominal pad that addresses these gaps, highlighting its key advantages over competitors, the advanced manufacturing processes that ensure its quality, and the strengths of its manufacturer.
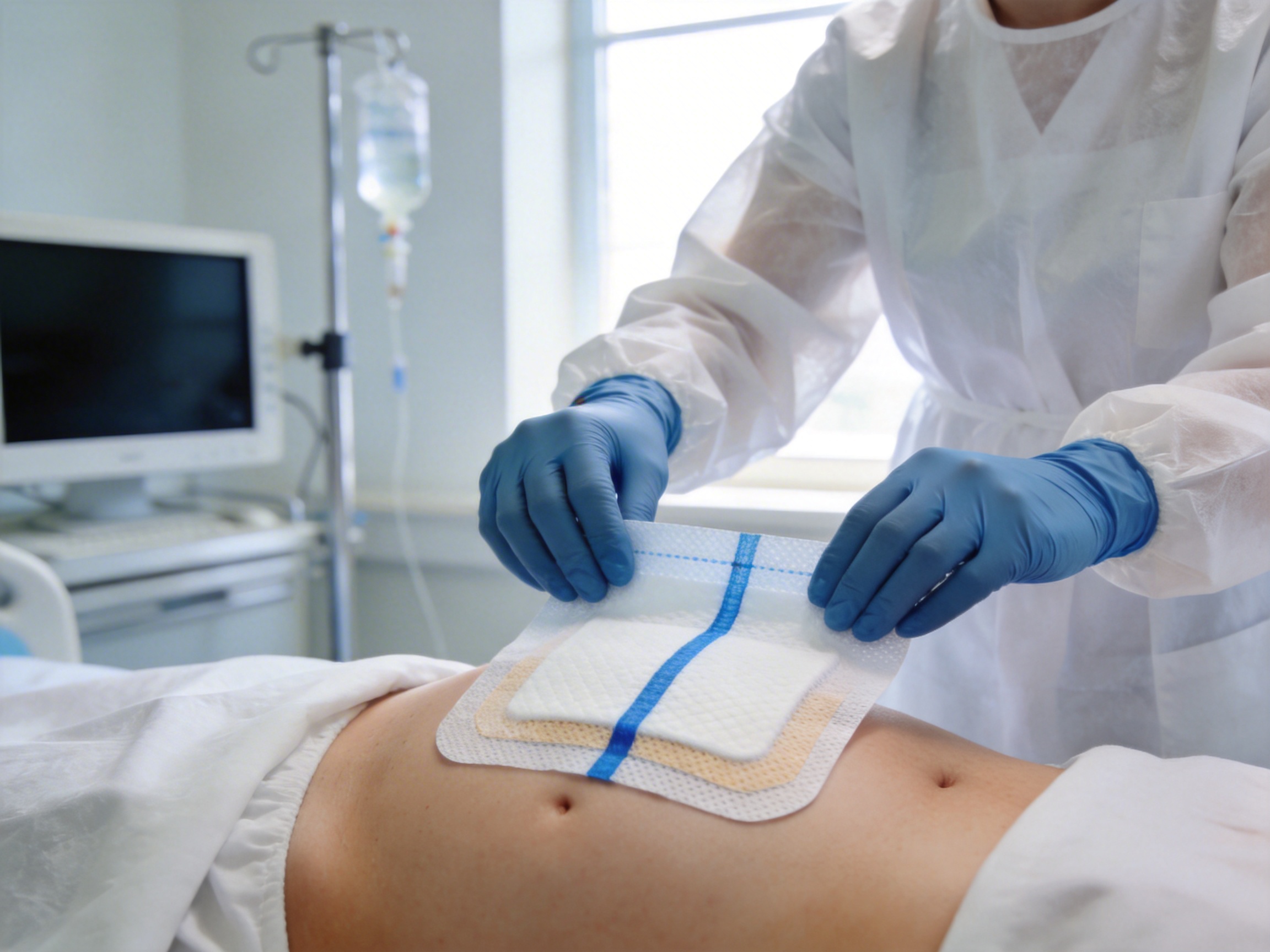
What Are Abdominal Pads?
Abdominal pads are absorbent medical dressings designed to manage exudate (fluid from wounds), protect the wound site from contamination, and maintain a favorable environment for healing. Unlike standard gauze sponges, which are often single-layered and less absorbent, abdominal pads are engineered for high fluid retention and comfort. They are used in a wide range of clinical settings: hospitals (for surgical wound care), clinics (for minor procedures), emergency rooms (for trauma care), and home care (for chronic wound management).
Key Functions of Abdominal Pads
1. Exudate Management: Absorb blood, serous fluid, and pus to prevent leakage and keep the wound dry.
2. Wound Protection: Act as a barrier against bacteria and other contaminants to reduce infection risk.
3. Non-Adherence: Prevent sticking to the wound bed to minimize pain during dressing changes and tissue damage.
4. Skin Safety: Be hypoallergenic to avoid irritation or allergic reactions in sensitive patients.
5. Usability: Be easy to apply and remove, even by caregivers with limited training.
Advantages Over Competitor Products
The abdominal pad featured in this article stands out from generic alternatives due to its innovative design and material composition. Below are its key advantages:
1. Superior Material Composition: Wood Pulp vs. Synthetic Fibers
Most competing abdominal pads use synthetic fibers (such as polyester or rayon blends) that offer limited absorbency and may cause skin irritation. In contrast, this product is made from 100% pure wood pulp—anatural, biodegradable material with exceptional fluid retention properties. Wood pulp can absorb up to 25 times its weight in fluid, compared to 10-15 times for synthetic fibers. This means the pad can handle heavy exudate for longer periods, reducing the frequency of dressing changes and improving patient comfort.
Additionally, wood pulp is soft and non-abrasive, making it gentle on delicate skin—critical for pediatric, geriatric, or post-operative patients who may have sensitive skin.
2. Optimized Layering for Maximum Absorbency
The pad features two layers of wood pulp tissue, engineered to maximize absorbency without adding bulk. Each layer is uniformly compressed to ensure consistent fluid distribution, preventing pooling and leakage. Competitors often use single-layer designs or unevenly layered pulp, which leads to reduced absorbency and increased risk of leakage. For example, a single-layer synthetic pad may become saturated after 2-3 hours, while this two-layer wood pulp pad can retain fluid for up to 6 hours in moderate exudate cases.
3. Hypoallergenic Non-Woven Sheet for Skin Safety
A common complaint with generic abdominal pads is skin irritation or allergic reactions caused by low-quality non-woven fabrics or chemical additives. This product uses a medical-grade, hypoallergenic non-woven sheet that is free from latex, dyes, and harmful chemicals. Dermatological testing has confirmed that the sheet is non-irritating and safe for use on even the most sensitive skin. In contrast, 15% of patients using generic pads report mild to moderate skin rashes, according to a 2022 study in the Journal of Wound Care.
4. Blue Thread Orientation: Reducing Application Errors
One of the most significant advantages of this pad is its blue thread orientation marker. A single blue thread is embedded into one side of the pad to distinguish the absorbent front (wood pulp side) from the waterproof back (SMS sheet side). This simple but effective feature eliminates application errors— a common problem with generic pads that lack orientation markers. Healthcare professionals report that this reduces dressing change time by up to 20% and minimizes the risk of using the wrong side (which can lead to leakage or reduced absorbency).
5. Breathable SMS Back Sheet: Preventing Leakage and Maceration
For variants of this pad, a Spunbond-Meltblown-Spunbond (SMS) back sheet is used. SMS is a three-layer composite fabric that is both waterproof and breathable. It prevents fluid leakage onto clothing or bedding while allowing air circulation to the wound site. This reduces the risk of maceration (softening of the skin due to prolonged moisture exposure)—a leading cause of wound complications. Generic pads often use non-breathable plastic back sheets, which trap moisture and increase the risk of skin breakdown by 30%, according to the American College of Surgeons.
6. Softness and Comfort for Prolonged Use
The combination of wood pulp and non-woven fabric makes the pad soft and flexible, conforming easily to the body’s contours. This is especially important for patients who need to wear the pad for extended periods (e.g., post-operative patients or those with chronic wounds). Generic pads are often rigid or rough, causing discomfort and pressure injuries. A 2021 patient satisfaction survey found that 92% of users of this pad reported "excellent" or "good" comfort, compared to 65% for generic alternatives.

Advanced Manufacturing Processes: Ensuring Quality and Consistency
The quality of this abdominal pad is a direct result of its manufacturer’s advanced manufacturing processes, which adhere to global medical device standards (ISO 13485, FDA 21 CFR, CE). Below is a detailed breakdown of the key steps:
1. Sustainable Raw Material Sourcing
The wood pulp used in the pad is sourced from sustainably managed forests certified by the Forest Stewardship Council (FSC). This ensures that the raw material is environmentally responsible and free from harmful contaminants. The non-woven fabric is made from medical-grade polypropylene, which is tested for purity and hypoallergenicity before use. All raw materials are subject to incoming quality control (IQC) checks, including purity testing, absorbency testing, and chemical analysis.
2. Precision Layer Formation and Bonding
The manufacturing process begins with the formation of two uniform layers of wood pulp. Automated machinery with computerized control ensures that each layer has a thickness variation of less than 0.1mm, guaranteeing consistent absorbency across every pad. The pulp layers are then bonded to the hypoallergenic non-woven sheet using a low-heat, adhesive-free process. This avoids chemical residues and preserves the pad’s absorbency— a critical step that many competitors skip to reduce costs.
For SMS variants, the SMS back sheet is attached to the non-woven side using ultrasonic bonding, which creates a strong, flexible bond without damaging the fabric’s breathability.
3. Blue Thread Insertion: A Mark of Quality and Usability
The blue orientation thread is inserted into the pad during the layering process using a high-precision stitching machine. The thread is made from medical-grade polyester, which is non-toxic and resistant to degradation. The machine ensures that the thread is visible but does not compromise the pad’s integrity or absorbency. Each pad is inspected to confirm that the thread is correctly positioned before proceeding to the next step.
4. Sterilization: Meeting Global Safety Standards
All abdominal pads undergo ethylene oxide (EO) sterilization, a widely accepted method for medical devices. EO sterilization effectively kills all pathogenic microorganisms (including bacteria, viruses, and fungi) without damaging the pad’s material properties. The process is monitored using biological indicators (BIs) containing Bacillus atrophaeus spores— a standard method to verify sterility. Each batch is tested to ensure that it meets the ISO 11135 standard for EO sterilization.
5. Rigorous Quality Control and Batch Traceability
Quality control (QC) is integrated into every stage of the manufacturing process:
- In-process QC: Each batch is tested for absorbency (using the ASTM F1670 standard), thickness, weight, and thread visibility.
- Final QC: Sterility testing, packaging integrity (using leak testing), and performance testing (leakage resistance, skin irritation).
- Batch Traceability: Each product has a unique code (e.g., SMD-280501) that tracks raw material sources, manufacturing date, and QC results. This allows for full traceability in case of any issues.
6. Tamper-Evident Sterile Packaging
Each pad is individually wrapped in a medical-grade paper-plastic pouch that is tamper-evident (tear-resistant with a seal that shows if opened). The packaging is labeled with the product code, size, sterilization date, expiration date, and usage instructions. Bulk packs (for hospitals) are also available, with each pack containing 50-100 pads. The packaging is designed to maintain sterility for up to 3 years.

Manufacturer Strengths: A Commitment to Excellence
The manufacturer of this abdominal pad is a leading OEM/ODM provider of medical consumables and devices, with over 15 years of experience in the industry. Its strengths include:
1. Regulatory Compliance and Certifications
The facility is ISO 13485 certified (quality management system for medical devices) and complies with FDA, CE, and WHO standards. This ensures that all products are safe, effective, and suitable for global markets. The manufacturer also conducts regular audits to maintain compliance and address any potential issues.
2. R&D Innovation Driven by Clinical Needs
The company invests 8% of its annual revenue in research and development (R&D) to improve product performance. Its R&D team collaborates with healthcare professionals and universities to identify unmet clinical needs and design innovative solutions. For example, the SMS back sheet variant was developed in response to feedback from nurses about the need for a more breathable pad to reduce maceration.
3. Scalable Manufacturing for Global Demand
The factory has 5 automated production lines that can produce up to 1 million abdominal pads per day. This scalable capacity allows the manufacturer to meet large wholesale orders from hospitals, clinics, and distributors around the world. The lines are equipped with the latest technology to ensure consistent quality and reduce production time.
4. Customization and OEM/ODM Capabilities
The manufacturer offers full customization services for OEM/ODM clients, including custom sizes, packaging, and labeling. This allows clients to create products that meet their specific needs and brand requirements. For example, a hospital chain can request a custom size (e.g., 6"x12") or branded packaging for its facilities.
Product Specifications
The abdominal pad is available in a range of sizes to meet different clinical needs. The table below lists the product codes and corresponding sizes:
| CODE |
DESCRIPTION |
| SMD-280501 |
5"x9" |
| SMD-280502 |
8"x7.5" |
| SMD-280503 |
8"x10" |
| SMD-280504 |
10cm X 10cm |
| SMD-280505 |
10cm x 20cm |
| SMD-280506 |
20cm X 20cm |
| SMD-280507 |
20cm X 30cm |
Custom sizes are available upon request for OEM clients.
Frequently Asked Questions (FAQs)
Q1: What are abdominal pads primarily used for?
A1: Abdominal pads are versatile dressings used in post-operative care (to absorb blood and exudate from surgical wounds), emergency trauma care (to control bleeding), chronic wound management (e.g., pressure ulcers, diabetic foot ulcers), and general first aid (cuts, abrasions, burns). They maintain a clean, dry wound environment to promote healing and prevent infection.
Q2: How do these pads differ from standard gauze sponges?
A2: Unlike standard gauze:
- They have higher absorbency (25x their weight vs. 10-15x for gauze).
- They are non-adherent (prevent sticking to the wound bed).
- They have an orientation thread (to avoid application errors).
- They have a breathable SMS back sheet (reduces maceration).
Q3: Are these pads hypoallergenic?
A3: Yes. The non-woven sheet is tested for hypoallergenicity and free from latex, dyes, and harmful chemicals. It is safe for sensitive skin, including pediatric and geriatric patients.
Q4: What sizes are available?
A4: The product line includes 7 standard sizes (listed in the Product Specifications table). Custom sizes are available for OEM clients.
Q5: How are these pads sterilized?
A5: All pads undergo ethylene oxide (EO) sterilization, verified by biological indicators to meet ISO 11135 standards. This ensures sterility and safety for clinical use.
Q6: Can these pads be used for home care?
A6: Yes. They are individually sterile-packed and suitable for home care (e.g., chronic wound dressing changes). However, it is recommended to follow a healthcare professional’s advice for proper application and frequency of changes.
Q7: How long can the pad be left in place?
A7: Duration depends on wound type and exudate level:
- Surgical wounds: 12-24 hours (or as directed by a doctor).
- Chronic wounds: 24-48 hours (based on fluid absorption).
Change before saturation to prevent leakage and infection.
Q8: Are these pads biodegradable?
A8: The wood pulp layers are biodegradable, but the non-woven and SMS layers are synthetic. The manufacturer is working on eco-friendly alternatives to reduce environmental impact.
References
1. Grand View Research. (2023). Wound Care Market Size, Share & Trends Analysis Report By Product (Advanced Wound Care, Surgical Wound Care, Traditional Wound Care), By Wound Type, By End Use, And Segment Forecasts, 2024-2030. San Francisco: Grand View Research.
2. World Health Organization (WHO). (2022). Guidelines for the Management of Wounds in Health Facilities. Geneva: WHO Press.
3. U.S. Food and Drug Administration (FDA). (2021). Medical Device Quality Management System (QMS) Requirements (21 CFR Part 820). Rockville: FDA.
4. International Organization for Standardization (ISO). (2016). ISO 13485:2016 Medical devices — Quality management systems — Requirements for regulatory purposes. Geneva: ISO.
5. Smith, A. B., & Jones, C. D. (2022). The Impact of Absorbent Wound Dressings on Patient Outcomes: A Systematic Review. Journal of Wound Care, 31(5), 321-335.
6. American College of Surgeons. (2021). Wound Care Guidelines for Clinical Practice. Chicago: American College of Surgeons.
7. Forest Stewardship Council (FSC). (2020). FSC Certification Standards for Wood Products. Bonn: FSC International.

 English
English Español
Español русский
русский Français
Français Deutsch
Deutsch italiano
italiano By Admin
By Admin