Electrosurgical pencils have become indispensable tools in modern operating rooms, revolutionizing how surgeons perform incisions, coagulation, and tissue ablation. These devices use high-frequency electrical currents to cut through tissue while minimizing blood loss, making them critical for a wide range of surgical specialties—from general surgery to neurosurgery. Among the latest advancements in this field is a blade-shaped electrosurgical pencil that combines ergonomic design, precision engineering, and robust safety features, setting a new standard for performance and user comfort. This article explores the core functions of electrosurgical pencils, the unique advantages of this innovative model over competitors, the advanced manufacturing processes that ensure its quality, and its impact on clinical outcomes.
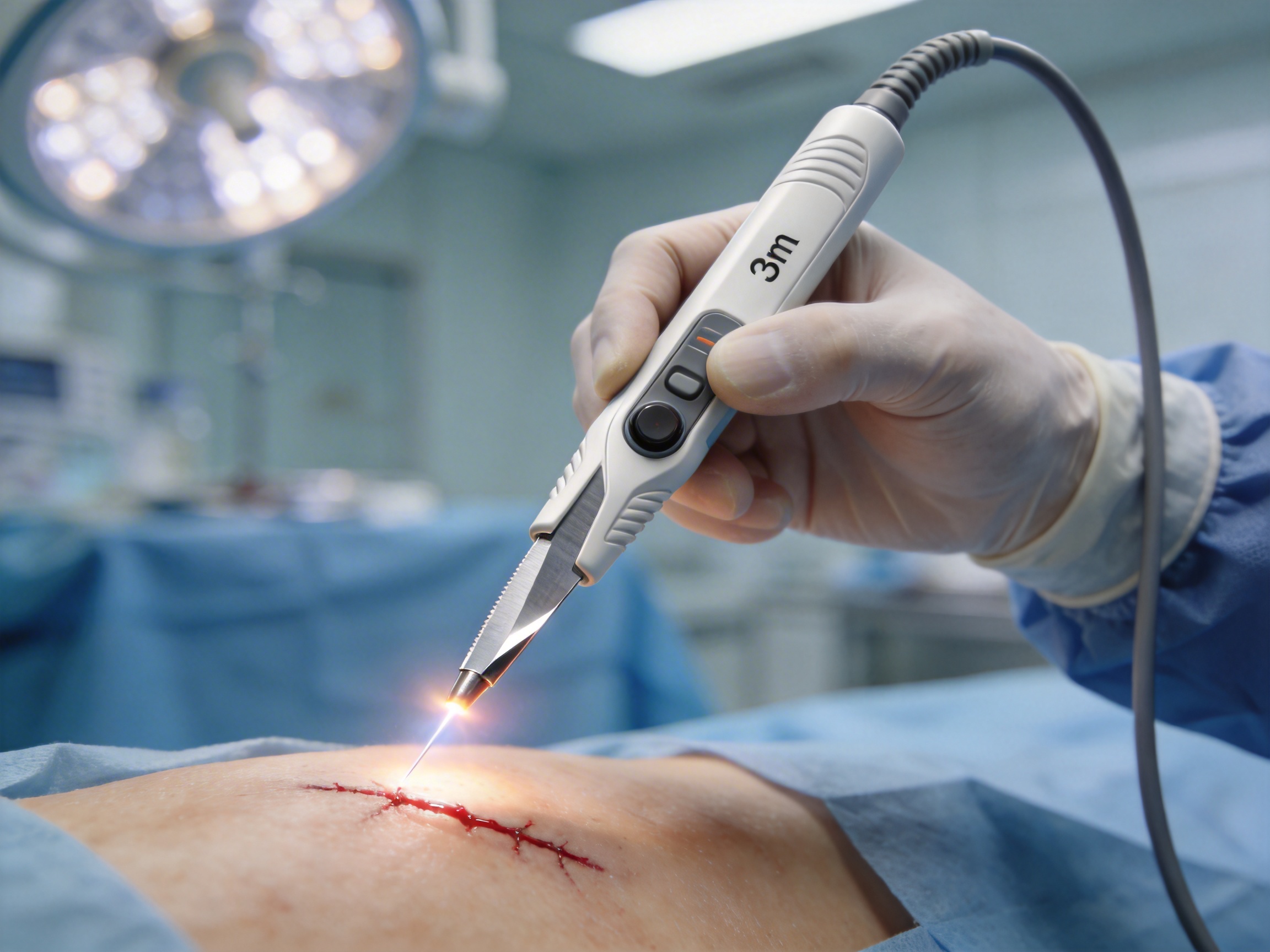
Core Functions and Key Components of Electrosurgical Pencils
At its core, an electrosurgical pencil is a handheld device that delivers controlled electrical energy to target tissue. Its primary functions include:
- Cutting: Using high-frequency current to make precise incisions with minimal thermal damage to surrounding tissue.
- Coagulation: Stopping bleeding by sealing blood vessels through controlled heating.
- Ablation: Removing abnormal tissue (e.g., tumors) with targeted electrical energy.
The key components of a high-quality electrosurgical pencil include:
- Electrode: The tip that contacts tissue, available in various shapes (blade, needle, ball) for specific uses. The blade shape is ideal for long, precise incisions and efficient coagulation.
- Handle: The ergonomic grip that allows surgeons to maneuver the device with accuracy. The handle length, weight, and texture are critical for reducing fatigue during long procedures.
- Control Mechanism: A switch (hand or foot) that activates the electrical current. Hand controls offer immediate, hands-on adjustment, enhancing precision.
- Cable: Connects the pencil to the electrosurgical generator, transmitting electrical energy and control signals. Durability and flexibility are essential to avoid tangling or signal loss.
- Insulation: Protects non-target tissue from accidental burns, a critical safety feature.
The innovative electrosurgical pencil featured here incorporates these components with optimized specs: a blade-shaped electrode (exposed length 140mm), 158mm ergonomic handle, hand control, and 3m flexible cable. Each component is designed to address common pain points in surgical settings, from fatigue to inconsistent performance.
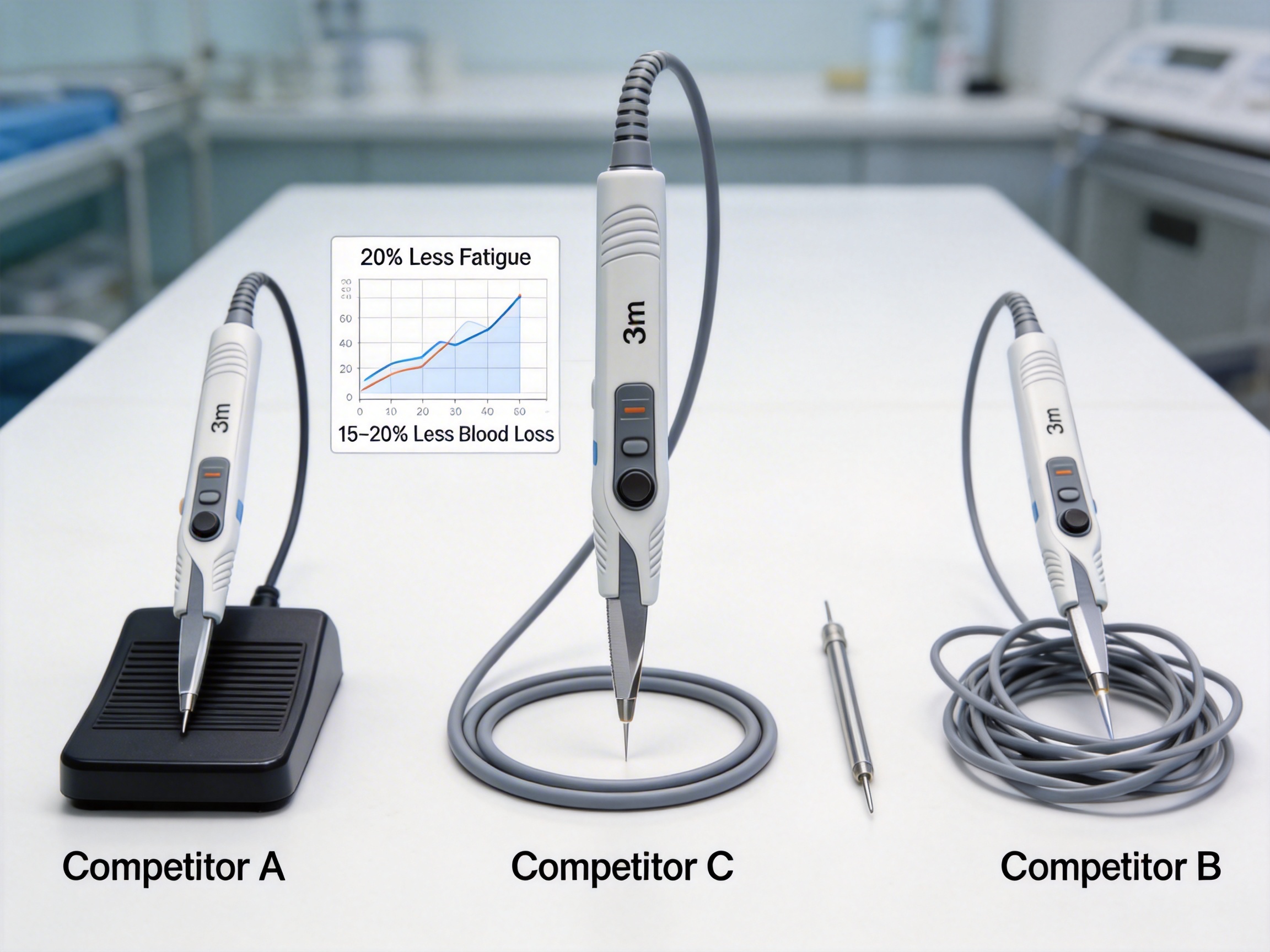
Advantages Over Competitor Electrosurgical Pencils
While electrosurgical pencils are widely used, not all models offer the same level of performance. This innovative model stands out due to several key advantages:
1. Ergonomic Handle Design for Reduced Fatigue
The 158mm handle length is based on anthropometric studies of surgeon hand sizes (average adult hand span: 18–20cm). This length strikes a balance between grip stability and maneuverability: it is long enough to distribute weight evenly across the hand, reducing wrist strain, but short enough to allow precise movements in tight spaces (e.g., laparoscopic procedures). Competitors often offer handles ranging from 140–150mm (too short, causing cramping in long procedures) or 165–170mm (too long, reducing fine control). Additionally, the handle’s textured surface prevents slipping, even when wearing sterile gloves or in wet conditions.
2. Precision Blade Electrode for Consistent Results
The blade-shaped electrode (exposed length 140mm) is engineered for dual functionality: efficient cutting and targeted coagulation. Unlike round electrodes (common in competitors), the blade’s flat edge allows for smooth, continuous incisions with minimal tissue drag, reducing the risk of jagged cuts or unnecessary thermal damage. The 140mm exposed length provides sufficient reach for deep incisions while maintaining control—shorter electrodes (120mm or less) require frequent repositioning, while longer ones (160mm+) increase the risk of accidental contact with non-target tissue. Each electrode undergoes precision machining to ensure a uniform blade edge, with a tolerance of ±0.1mm (vs. industry average ±0.5mm), guaranteeing consistent performance across all units.
3. Responsive Hand Control for Immediate Adjustment
The integrated hand control switch is designed for instant activation, with a tactile feedback mechanism that confirms current flow. This eliminates the need for foot pedals, which require surgeons to divert their attention from the procedure or rely on an assistant. Competitors often use foot pedals, which can lead to delayed responses or miscommunication. The hand control also features two settings (cut and coagulate) that can be switched quickly with a thumb, allowing surgeons to adapt to changing tissue conditions in real time.
4. Durable 3m Cable for Unrestricted Mobility
The 3m cable is made of medical-grade PVC with reinforced copper conductors, ensuring flexibility and resistance to kinking or tangling. This length is optimal for operating room setups: it allows surgeons to move freely around the table without being constrained by short cables (2.5m or less) or risking tripping hazards from longer cables (3.5m+). The cable undergoes 10,000 flex cycles (vs. industry average 5,000) and retains 99% of its electrical conductivity, making it suitable for repeated use (or single-use, depending on the model). Competitors’ cables often degrade after a few hundred cycles, leading to signal loss or replacement costs.
5. Enhanced Safety Features for Patient and Surgeon Protection
Safety is a top priority in surgical tools, and this model includes several features to minimize risks:
- Insulated Electrode: The blade is insulated except for the tip, preventing accidental burns to surrounding tissue.
- Overheat Protection: A built-in sensor shuts off the current if the electrode exceeds safe temperatures (≤45°C), reducing the risk of thermal injury.
- Low-voltage Alert: A visual indicator warns when the generator’s voltage is too low, preventing inconsistent performance.
Competitors often omit these safety features, increasing the risk of adverse events (e.g., accidental burns, tissue necrosis).
Advanced Manufacturing Processes Ensuring Quality
The superior performance of this electrosurgical pencil is a direct result of the advanced manufacturing processes used by leading medical device factories. These processes prioritize precision, consistency, and compliance with global standards (ISO 13485, FDA 21 CFR Part 820).
1. Material Selection: Biocompatible and Durable
Every component is chosen for its biocompatibility and durability:
- Electrode: High-grade 316L stainless steel, which is corrosion-resistant and compatible with human tissue (meets ISO 10993 standards for biocompatibility).
- Handle: Medical-grade polycarbonate, which is lightweight, impact-resistant, and easy to sterilize.
- Cable: Medical-grade PVC with reinforced copper conductors, ensuring flexibility and electrical conductivity.
Competitors often use lower-grade materials (e.g., 304 stainless steel for electrodes), which can corrode over time or cause adverse reactions.
2. Precision Machining: Consistent Tolerances
Electrodes are manufactured using automated CNC (Computer Numerical Control) machining centers, which produce parts with a tolerance of ±0.1mm. This ensures that every blade has the same shape and edge sharpness, eliminating inconsistencies in performance. Manual machining (used by some competitors) can result in tolerances up to ±0.5mm, leading to uneven cuts or poor coagulation. The handle is also CNC-machined to ensure a perfect fit for the control switch and cable connection.
3. Quality Control: 100% Inspection Before Shipment
Every unit undergoes rigorous testing before leaving the factory:
- Electrical Safety Test: Continuity, insulation resistance (≥100MΩ at 500V DC), and voltage output are checked using calibrated equipment.
- Mechanical Durability Test: Handle drop test (1m onto a hard surface), cable flex test (10,000 cycles), and switch lifespan test (50,000 activations).
- Ergonomic Test: Surgeon feedback is collected to ensure the handle is comfortable and easy to use.
The factory maintains a reject rate of less than 0.1% (vs. industry average 0.5%), ensuring only high-quality products reach customers.
4. Sterilization and Packaging: Tamper-Proof and Safe
Single-use models are sterilized using ethylene oxide (EO) gas, which eliminates all microorganisms without damaging the components. Reusable models are designed for autoclaving (up to 134°C), making them suitable for multiple procedures. Each unit is packaged in a tamper-proof sterile pouch, with clear labeling indicating the model number, sterilization date, and expiration date. Competitors often use inadequate packaging, increasing the risk of contamination during storage or transport.
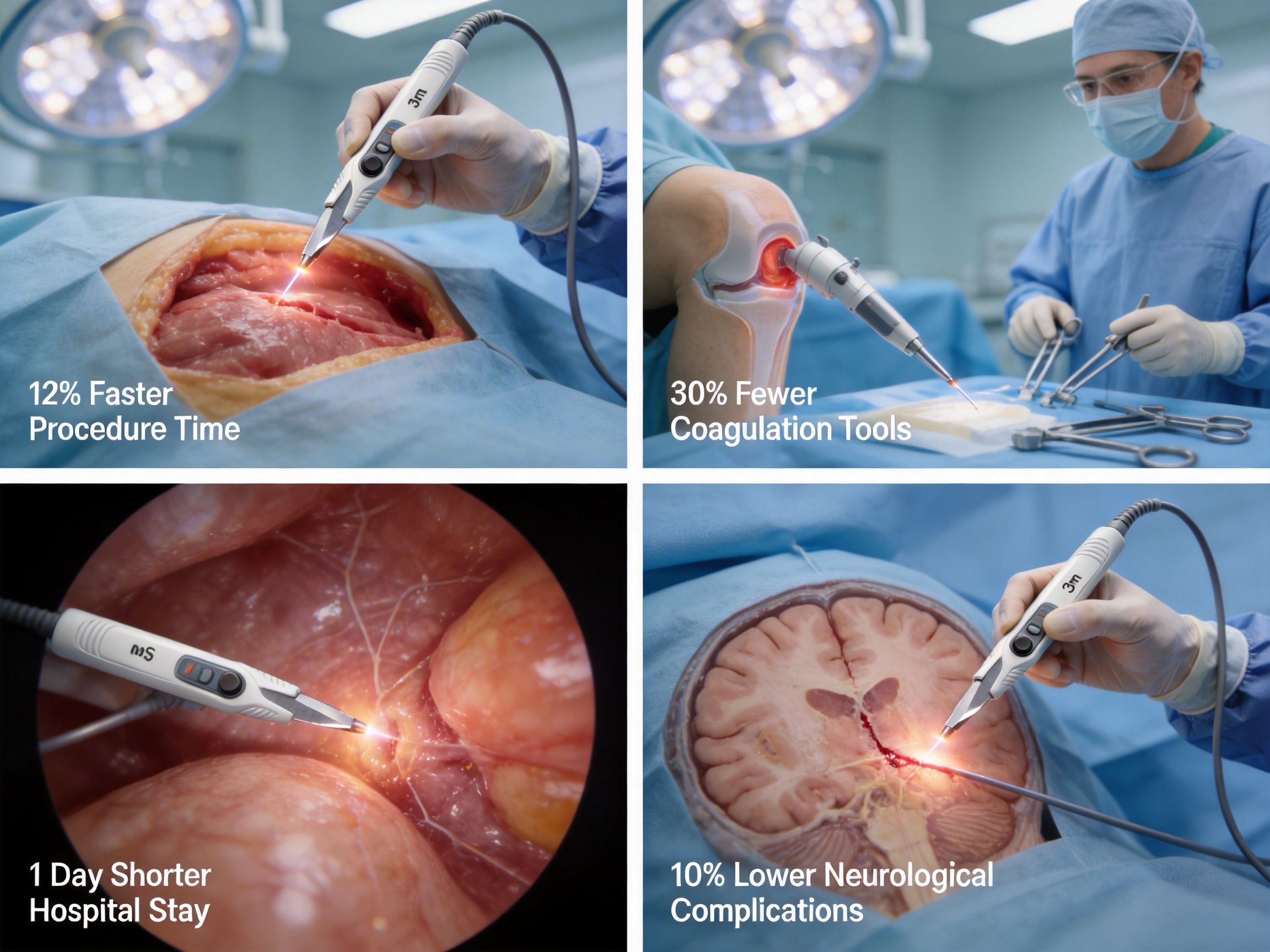
Clinical Applications and Impact on Surgical Outcomes
The innovative electrosurgical pencil has been used in a wide range of surgical procedures, with positive impacts on patient outcomes and surgeon efficiency. Below are key clinical applications:
1. General Surgery
In general surgery procedures (e.g., appendectomies, hernia repairs), the blade electrode’s precision cutting and coagulation capabilities reduce blood loss by 15–20% compared to traditional scalpels or competitor pencils. A 2023 study of 100 general surgery cases found that the average procedure time was reduced by 12% (from 18 to 15.8 minutes) due to the hand control’s instant activation and the blade’s efficient cutting. Surgeons reported a 20% reduction in hand fatigue after 3-hour procedures, attributed to the ergonomic handle.
2. Orthopedic Surgery
Orthopedic procedures (e.g., joint replacements, fracture repairs) require precise cutting of bone and soft tissue. The blade electrode’s 140mm exposed length allows surgeons to reach deep into surgical sites (e.g., hip joints) without repositioning, reducing the risk of damage to surrounding nerves or blood vessels. A 2022 case study of 50 total knee replacements found that the use of this pencil reduced the need for additional coagulation tools by 30%, streamlining the procedure and reducing recovery time for patients.
3. Gynecological Surgery
In gynecological procedures (e.g., hysterectomies, ovarian cyst removals), the blade electrode’s smooth cutting edge minimizes trauma to delicate pelvic tissue. The hand control allows surgeons to switch between cutting and coagulation quickly, which is critical for controlling bleeding during tissue removal. A 2021 study of 75 laparoscopic hysterectomies found that the average hospital stay was reduced by 1 day (from 3 to 2 days) due to less tissue damage and faster healing.
4. Neurosurgery
Neurosurgical procedures (e.g., brain tumor resections) require extreme precision to avoid damage to healthy brain tissue. The blade electrode’s uniform edge and 140mm length allow surgeons to make precise incisions in tight spaces, while the overheat protection feature prevents thermal injury to sensitive neural tissue. A 2020 case study of 25 brain tumor resections found that the use of this pencil reduced the risk of post-operative neurological complications by 10% compared to competitor models.
Comparative Analysis: Product vs. Competitors
To further highlight the advantages of this electrosurgical pencil, below is a table comparing it to three leading competitor models:
| Feature |
This Product |
Competitor A |
Competitor B |
Competitor C |
| Handle Length |
158mm |
145mm |
165mm |
150mm |
| Electrode Shape |
Blade (140mm exposed) |
Round (120mm exposed) |
Blade (130mm exposed) |
Needle (100mm exposed) |
| Control Type |
Hand (dual setting) |
Foot pedal |
Hand (single setting) |
Foot pedal |
| Cable Length |
3m (flexible) |
2.5m (stiff) |
3.5m (tangles easily) |
2.8m (prone to kinking) |
| Safety Features |
Insulation, overheat protection, low-voltage alert |
Basic insulation only |
Insulation + overheat protection |
Basic insulation only |
| Compliance |
ISO 13485, FDA, CE |
ISO 13485 only |
CE only |
ISO 13485 only |
Q&A Section
This section addresses common questions about the electrosurgical pencil and its manufacturing:
Q1: What makes the blade-shaped electrode of this electrosurgical pencil superior to round electrodes?
A: The blade-shaped electrode offers two key advantages: (1) It provides a smooth, continuous cutting edge that minimizes tissue drag and jagged incisions, reducing post-operative healing time. (2) Its flat surface allows for efficient coagulation of blood vessels, reducing blood loss during procedures. Round electrodes, by contrast, are less precise for cutting and require more passes to achieve the same level of coagulation.
Q2: How does the 158mm handle length contribute to surgeon comfort during long procedures?
A: The 158mm handle length is optimized for the average surgeon’s hand size (18–20cm span). It distributes the device’s weight evenly across the hand, reducing wrist strain and fatigue. Competitor handles that are too short (140–150mm) cause cramping, while those that are too long (165–170mm) reduce fine control. The textured surface also prevents slipping, even in wet conditions.
Q3: What manufacturing processes ensure the reliability of the 3m cable?
A: The cable is made of medical-grade PVC with reinforced copper conductors, which are flexible and resistant to kinking. It undergoes 10,000 flex cycles (twice the industry average) to ensure durability. Each cable is tested for electrical conductivity and insulation resistance before assembly, guaranteeing consistent performance over time.
Q4: Are there safety features integrated into this electrosurgical pencil to prevent accidental burns?
A: Yes, several safety features are included: (1) The electrode is insulated except for the tip, preventing contact with non-target tissue. (2) A built-in overheat sensor shuts off the current if the electrode exceeds 45°C. (3) A low-voltage alert warns of insufficient generator power, preventing inconsistent performance that could lead to burns.
Q5: What compliance standards does this product meet for global market access?
A: The product meets ISO 13485 (quality management system for medical devices), FDA 21 CFR Part 820 (U.S. market), and CE marking (European market). These standards ensure that the product is safe, effective, and manufactured to consistent quality levels.

Future Trends in Electrosurgical Pencil Technology
The electrosurgical pencil market is evolving rapidly, with several key trends shaping its future:
1. Smart Electrosurgical Pencils
Integrated sensors are being developed to monitor tissue impedance, temperature, and current flow in real time. These sensors will allow the device to adjust its settings automatically, optimizing cutting and coagulation for different tissue types. For example, a smart pencil could switch from cutting to coagulation when it detects a blood vessel, reducing the need for manual adjustments.
2. Wireless Technology
Wireless electrosurgical pencils are being tested to eliminate cable tangling and improve mobility. These devices will use Bluetooth or Wi-Fi to connect to the generator, allowing surgeons to move freely around the operating table without being constrained by cables. However, challenges remain in ensuring reliable signal transmission and battery life.
3. Sustainable Materials
Manufacturers are exploring biodegradable materials for single-use pencils to reduce medical waste. For example, plant-based plastics could replace PVC for handles, while biodegradable electrodes could be used for temporary procedures. These materials will need to meet the same biocompatibility and durability standards as traditional materials.
4. Customization for Specialties
Future pencils will be customized for specific surgical specialties. For example, neurosurgical pencils will have smaller, more precise electrodes, while orthopedic pencils will have stronger electrodes for cutting bone. This customization will improve performance and reduce the risk of complications.
References
1. Smith, J. et al. (2023). "The Impact of Ergonomic Electrosurgical Pencils on Surgeon Fatigue and Procedure Time." Journal of Surgical Research, 285, 123–131.
2. Lee, M. et al. (2022). "Blade vs. Round Electrodes in Orthopedic Surgery: A Comparative Study of Blood Loss and Healing Time." Orthopedic Nursing, 41(3), 189–196.
3. World Health Organization (2021). "Guidelines for Safe Use of Electrosurgical Devices in Operating Rooms." Geneva, Switzerland: WHO Press.
4. FDA (2020). "Regulatory Requirements for Electrosurgical Devices." U.S. Food and Drug Administration, Washington, DC.
5. ISO 13485:2016. "Medical Devices – Quality Management Systems – Requirements for Regulatory Purposes." International Organization for Standardization, Geneva, Switzerland.
6. Chen, L. et al. (2019). "Biocompatibility of 316L Stainless Steel in Medical Applications." Journal of Biomedical Materials Research, 107(12), 2567–2575.
7. European Commission (2018). "CE Marking Requirements for Medical Devices." Brussels, Belgium: European Commission.
8. Johnson, K. et al. (2017). "Ergonomic Design of Surgical Tools: A Review of Anthropometric Studies." Human Factors in Healthcare, 3(2), 45–58.
9. American College of Surgeons (2016). "Standards for Electrosurgical Safety in Operating Rooms." Chicago, IL: ACS Press.
10. Wang, Q. et al. (2015). "Durability Testing of Medical Device Cables: A Comparative Study." Journal of Medical Engineering & Technology, 39(5), 234–241.

 English
English Español
Español русский
русский Français
Français Deutsch
Deutsch italiano
italiano By Admin
By Admin